NEW YORK — It’s OK for doctors to start using a kid-friendly nasal spray flu vaccine again, a federal panel said Wednesday.
Two years ago, the advisory group pulled its recommendation for FluMist vaccine after research found it wasn’t working against swine flu, the kind of flu that was making most people sick then. But the Advisory Committee of Immunization Practices voted 12-2 Wednesday to recommend the nasal spray as an option for next winter’s flu season.
An official from AstraZeneca, the company that makes FluMist, said the problem with the vaccine has been identified and corrected. But panel members noted there’s still not good proof that FluMist works well against the swine flu bug.
“This is not an easy decision. It’s always a challenge to make a decision with incomplete data,” said one panel member, Dr. Edward Belongia of the Wisconsin-based Marshfield Clinic Research Foundation.
The panel makes its recommendations to the Centers for Disease Control and Prevention, which usually accepts the advice and sends it along as guidance to doctors, hospitals and health insurers.
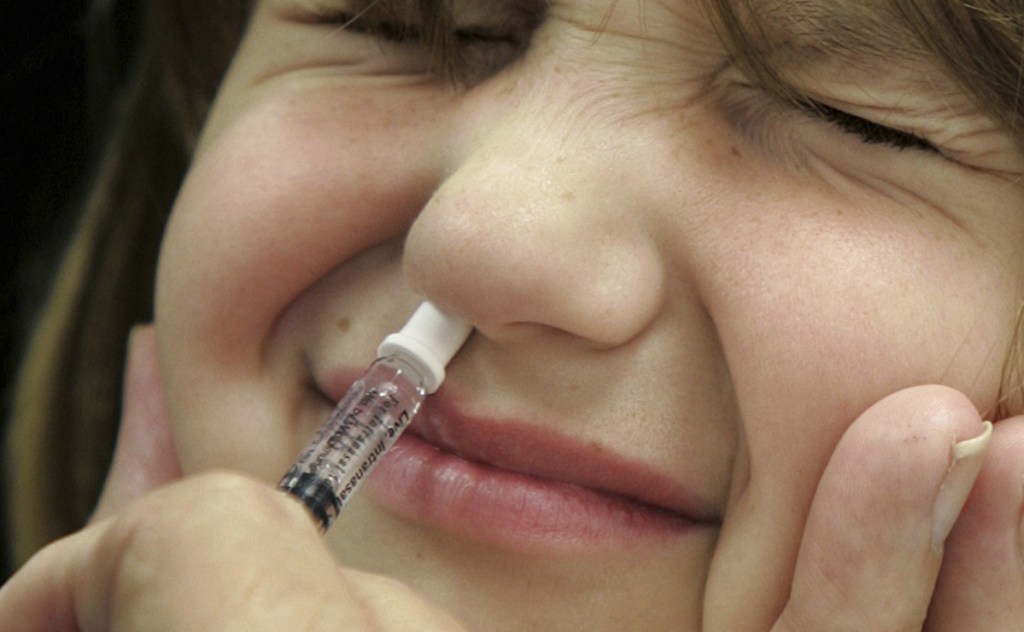
FluMist is the only spray-in-the-nose vaccine on the market. It was first licensed in 2003 and is approved for healthy people ages 2 to 49.
Send questions/comments to the editors.


Success. Please wait for the page to reload. If the page does not reload within 5 seconds, please refresh the page.
Enter your email and password to access comments.
Hi, to comment on stories you must . This profile is in addition to your subscription and website login.
Already have a commenting profile? .
Invalid username/password.
Please check your email to confirm and complete your registration.
Only subscribers are eligible to post comments. Please subscribe or login first for digital access. Here’s why.
Use the form below to reset your password. When you've submitted your account email, we will send an email with a reset code.