Portland-based ImmuCell Corp. said Tuesday that it plans to seek $3.5 million in private investment to help fund a $20 million production facility for a new animal health product for which the company is seeking U.S. Food and Drug Administration approval.
ImmuCell also released a preliminary earnings estimate for the third quarter, in which product sales were down 20 percent from a year earlier.
Maine’s smallest publicly traded company, which makes products that improve bovine health and productivity in the dairy and beef industries, said it has entered into agreements with institutional and accredited investors for the private placement of roughly $3.5 million of ImmuCell’s common stock.
ImmuCell said in a news release that it has agreed to sell nearly 660,000 shares at $5.25 per share to outside investors. ImmuCell stock, which trades on the Nasdaq exchange under the symbol ICCC, was trading at $7 a share when the market opened Tuesday.
“This new capital, plus cash flow from operations and possibly some incremental bank debt, fully funds our $20 million capital expenditure budget for the construction of our pharmaceutical facility to produce Nisin, the active ingredient in Mast Out,” said ImmuCell President and CEO Michael Brigham in the release. “We look forward to completing this offering, which will supplement the offering we completed earlier in the year.”
In February, the company completed a $5.9 million public offering in which it offered for sale 1.1 million shares of stock, also priced at $5.25 each. The offering diluted the value of ImmuCell’s existing pool of 3.1 million outstanding shares by roughly one-third.
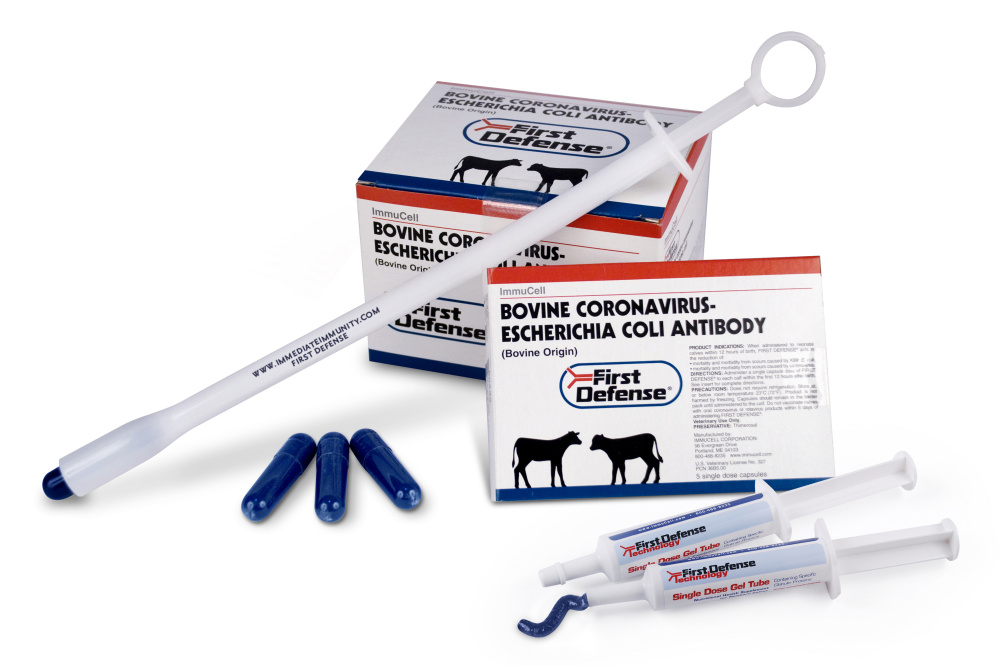
Mast Out is a novel treatment for dairy cows with mastitis, an inflammation of the udders that is often caused by a bacterial infection. Existing mastitis treatments require dairy farmers to throw away the milk for a certain time period following treatment, but Mast Out would not. The product has not yet received FDA approval, and building a production facility for its key ingredient, Nisin, is one of the prerequisites. Nisin is a preservative with antibacterial properties that is made from milk and used in processed cheeses, meats and other products.
Also on Tuesday, ImmuCell released a preliminary earnings estimate for the third quarter. It estimated sales of $2 million for its flagship product First Defense during the quarter, a 20 percent decrease from sales of $2.5 million a year earlier.
The company explained that while it completed an expansion in early 2016 to double capacity and eliminate an 18-month backlog of orders, some customers moved on to competing products because of the wait time to obtain First Defense. The product boosts immunity to scours, or diarrhea, in newborn calves and must be administered within a few days of birth.
ImmuCell also said a competing product that had been off the market in 2014 and the first half of 2015 returned, and that some customers switched back to that product.
ImmuCell estimated that its net income for the quarter could range anywhere from a slight profit of $33,000 to a loss of $67,000, down from a profit of $351,000 in the third quarter of 2015. The company’s official earnings report is expected to be released Nov. 10.
After receiving approval for $375,000 in tax increment financing from the Portland City Council in September, ImmuCell began construction of its Nisin production facility on Oct. 11. As of Sept. 30, the end of the third quarter, ImmuCell had spent about $650,000 on the project, which contributed to its relatively flat estimated earnings along with the reduced sales of First Defense.
“The decrease in sales will correspondingly affect our profitability for the quarter,” Brigham said.
Copy the Story LinkSend questions/comments to the editors.





Success. Please wait for the page to reload. If the page does not reload within 5 seconds, please refresh the page.
Enter your email and password to access comments.
Hi, to comment on stories you must . This profile is in addition to your subscription and website login.
Already have a commenting profile? .
Invalid username/password.
Please check your email to confirm and complete your registration.
Only subscribers are eligible to post comments. Please subscribe or login first for digital access. Here’s why.
Use the form below to reset your password. When you've submitted your account email, we will send an email with a reset code.